|
Draw the molecular orbital diagram of dioxygen and calculate bond order. Reason : Electrons are removed from the antibonding molecular orbital from H2. 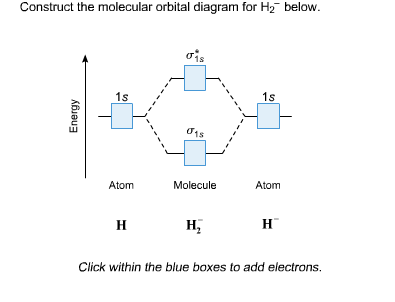
Bond order 1 2 ( number of bonding electron - number of antibonding electrons) 1 2 ( 10 - 4) 1 2 × 6 3. In the molecular orbital diagram for the molecular ion, N + 2, the number of electrons in the 2p molecular orbital is : Q. 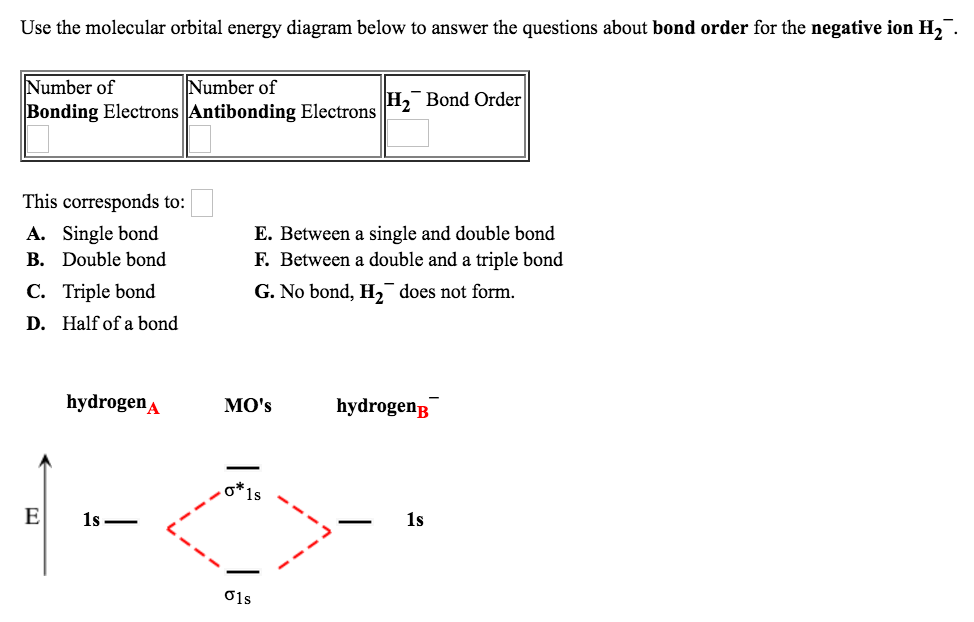
In other words, when the H-H bond forms, each atom loses heat energy equal to 2 x E. The electrons are more stable, lower energy, in the molecular orbital than they were in the separated atomic orbitals. 
It shows electrons in both bonding and anti-bonding molecular orbital. These 2 electrons go to fill the lowest energy molecular orbital, the sigma bonding orbital. In a sigma star (σ*) orbital An antibonding molecular orbital in which there is a region of zero electron probability (a nodal plane) perpendicular to the internuclear axis. The molecular orbital configurations of Li2, Be2, B2 and C2 molecules along with the bond order and magnetic properties are given below. The molecular orbital diagram has molecular orbital energy level at centre and is surrounded by atomic orbital energy level. Make a molecular orbital diagram for SO2 using group theory Consider the Molecular Orbital Diagram of N2+ and answer the following questions: Write a correct molecular orbital diagram for each of the following. Describe the hydrogen molecule in light of the following: HH H H.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
